All published articles of this journal are available on ScienceDirect.
In vivo Anti-Plasmodium Activity and Toxicity of Afzelia bipindensis and Senna Siamea Extracts: A Murine Model
Abstract
Background:
Malaria, a parasitosis affecting man, remains a public health problem in developing countries where morbidity and mortality are very high. Afzelia bipindensis and Senna siamea are two plants used in the treatment of malaria in different African countries including Cameroon.
Objective:
The aim of the present study was to evaluate the antiplasmodial activity of hydroethanolic leaves extracts of Afzelia bipindensis and Senna siamea, from Northern Cameron using Plasmodium berghei and to investigate the acute and sub-acute toxicity of leaves extracts in a rodent model.
Methods:
The four days Peter’s suppressive test was used to evaluate the antiplasmodial activity and the OCDE 423 and 412 guidelines were applied to evaluate acute and sub-acute toxicity. Biochemical tests related to hepatic, cardiac and renal toxicity were also assessed.
Results:
The leaves’ extracts of Afzelia bipindensis at doses 180, 360, 720 mg/kg and Senna siamea at doses of 100, 200, 400 mg/kg have shown significant antiplasmodial activity (P) with parasite reduction ≈ 50%. No mortality of rats was observed at the tested doses. The biochemical analysis did not reveal any statistically significant difference when compared with control. However, ALT was statistically increased at a higher dose (720 mg/kg) of Afzelia bipindensis leaves extract. On the other hand, there was a significant decrease in triglycerides at 360 mg/kg and 720 mg/kg
Conclusion:
It is concluded that daily consumption of leaves extract of Afzelia bipindensis and Senna siamea are without significant risks to human health, favoring the use of these products in the treatment of malaria.
1. INTRODUCTION
Malaria is a parasitic disease widely distributed around the world. This parasitosis is a handicap to the development of many countries. Every year, about 250 million people become sick and nearly 1 million die [1]. In 2000, malaria transmission was active in 106 countries and territories with 262 million cases and 839,000 fatalities. In 2016, malaria was active in 95 countries with 214 million cases worldwide and 438,000 deaths, among which 188 million cases were recorded in Africa with 395,000 deaths. Sub-Saharan Africa is the continent most affected by this epidemic with about 88% of cases and deaths worldwide [2]. In Cameroon, malaria is very dangerous and remains one of the main causes of morbidity and mortality. In 2016, the mortality rate caused by malaria was still high with nearly 4,000 registered deaths and the most affected areas were the Far North and North with 40% of deaths [3]. The main epidemiological types of malaria in Africa are found in Cameroon and today the fight against this epidemic remains oriented towards the vector agent, the curative and prophylactic treatment. In order to face such a problem in the world, control strategies have been developed such as awareness of the population, the vector control, and the prevention of malaria in pregnant women and the early and effective treatment of cases [4]. WHO has reported a dramatic decline in malaria over the last 15 years. Despite this remarkable progress, much remains to be done as malaria continued to decimate African populations with considerable loss of human life. The Global Technical Strategy for Malaria 2016-2030 has set ambitious and achievable targets for 2030, including a reduction of at least 90% in malaria incidence and mortality. The chemo-therapeutic treatment of malaria has evolved over the past 10 years as a result of the spread of multidrug-resistant strains of Plasmodium falciparum, and WHO has now promoted combination therapies based on artemisinin and or its derivatives (ACT) as Reference medicine for the management of uncomplicated malaria in order to reduce the risk of resistance [5] However, some signs of resistance to artemisinin have recently been detected in Asia, posing a huge threat to the control of malaria Disease [6]. The discovery of new antimalarial drugs is urgently needed, and plants were believed to play an important role in this new challenge, which is why current strategies are guiding research into the research and development of new antimalarial drugs. The plants of the traditional pharmacopoeia constitute an interesting research material because the main antimalarial drug currently used in therapeutics (quinine and artemisinin) remain predominantly of plant origin. Although, uncontrolled use of these plants may lead to other health complications [7-9]. According to WHO, 80% of rural people living in developing countries depend on traditional medicine for their primary health care requirement. The art of healing by plants has been known and practiced in Africa for a very long time because it exploits knowledge and transmitted orally from generation to generation to certain categories of initiated individuals that are the traditional healers and herbalists. Medicinal plants and knowledge about medicinal plants and traditional medicines are an important heritage of the African continent. Moreover, for the vast majority of the world's population, traditional medicine is the only treatment opportunity, while for many others, it is a choice motivated by a range of reasons that vary from place to place, from culture to person.
Our objective was to evaluate the antiplasmodial activity of hydroethanolic leaves extracts of Afzelia bipindensis and of Senna siamea harvested in Northern Cameroon. The outcome of the study provide an effective alternative for the treatment of malaria.
2. MATERIALS AND METHODS
2.1. Leaves of A. bipindendis and S. siamea
A. bipindensis and S. siamea are plants that are cultivated in Africa. A. bipindensis is a tree whereas the S. siamea is a legume that’s routinely used as food in Africa. The leaves of A. bipindensis Fig. (1A) and S. siamea Fig. (1B) were harvested during the month of December 2017 in the city of Maroua, Cameroon. After harvest, these leaves were dried for one week in the dark at room temperature, then crushed in mortars and sieved to obtain a powder with good particle size.

2.2. Extract Preparation
Extracts were prepared according to Zirihi et al. [10]. The leaves of Afzelia bipindensis and Senna siamea were kept in a dry area, away from the sun. They were separately crushed in a grinding mill. Hundred grams of each powder obtained was introduced in 1.5L of ethanol 70%. Each mixture was homogenised for several minutes using a clean wooden stick and left to stand for 24h. Then the gross macerates obtained were separated from the residual powder by pressing, then filtering through a Whatman No. 1 filter paper. The filtrates obtained were concentrated in a rotavapor then evaporated in an oven for 2 days at 40°C. Pasty and darkish hydroethanolic extracts for Senna siamea and greenish for Afzelia bipindensis were obtained.
2.3. Phytochemical Analysis and Preparation of Test Solutions
The phytochemical analysis was carried as previously described by Harborne [11]. Based on the color intensity of the solutions, five cutoffs; absence (-), trace (+/-), low (+), abundant (++) and very abundant (+++) were used to define the intensity of each metabolite in the plants.
The extract (100g) was dissolved in 0.5L distilled water, and the mixture was homogenized by agitation (5-10 min) using a magnetic stirrer. The solutions obtained were kept in a closed plastic jar and stored at 4oC after every administration session to animals. The volumes of the solutions administered to animals were calculated using the following formula.
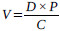
|
Where; V = volume of solution administered (mL); D = dose (mg/kg); P = animals weight (Kg); C = concentration of the solution to be administered (mg / mL).
2.4. In vivo Evaluation of the Antiplasmodial Activity of Afzelia bipindensis and Senna siamea Extracts in BALBc Mice
The antiplasmodial activity was evaluated according to the method described by Trager and Jensen 1976 [12]. The experimental mice (n=30) were inoculated by intraperitoneal injection at day 0, with 0.2 ml of infected blood containing 1 x 107 Plasmodium berghei NK65 strain in blood. Three hours after inoculation of the parasite, the animals were divided into 5 groups of 6 animals (G1-G5). The first three groups (G1, G2, and G3) of BALBc mice received the hydroethanolic extracts daily for four days by oral gavage using an oesophageal intubation tube. Doses of 180, 360 and 720 mg/kg for Afzelia bipindensis and 100, 200, and 400 mg/kg for Senna siamea were chosen for the treatment. The fourth group (G4) of mice received 200 µl of water and the last group (G5) received quinine (10 mg/kg). Blood smears were prepared on day 5 by collecting a drop of blood from the animals’ tails on a glass slide, then fixed with methanol and stained with giemsa staining procedure. The parasite count was obtained by using the modified Zucker and Campbell [13] formula:
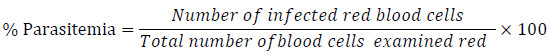
|
The efficiency of the treatment for each extract was evaluated by calculating the mean percentage of reduction of parasite according to the equation:
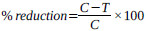
|
Where; C = Mean parasitemia percentage in the negative control group
T = mean parasitemia percentage in the treated group
2.5. Evaluation of Acute Toxicity
Acute toxicity of Afzelia bipindensis and Senna siamea extracts were evaluated in albino rats according to the OCDE no. 423 [14] guidelines for trials using chemical substances adopted on the 17 of December 2001. Four geometrically increasing doses of 2000 mg/kg, 3000 mg/kg, 4000 mg/kg, and 5000 mg/kg of the substance were administered by oesophageal intubation. The substance is tested in a sequential manner in which three same-sex animals were used at each stage. The animals were observed within 48 hours after administration of a dose, if all the animals survived, a higher dose was administered until the dose led to mortality.
2.6. Evaluation of Sub-acute Toxicity
Sub-acute toxicity of Afzelia bipindensis and Senna siamea was evaluated in rats according to the OCDE no. 412 [15] guidelines for trials using chemical products adopted on the 7th September 2009. Each group of rats received doses of 180, 360 or 720 mg/kg for Afzelia bipindensis and 100, 200 or 400 mg/kg orally for Senna siamea, using an intubation tube for 28 days. The control group received an equal volume of distilled water. The experimental albino rats were observed every day during the 28 days of treatment and weighed every week until the 28th day. Experimental animals were observed everyday morning and evening to see symptoms of morbidity and mortality. From the fourth week of exposition, the sensorial reactiveness to auditory and proprioceptive stimuli, prehension force and motor activity were also evaluated every day.
Weekly body weight variations were noted. The formula used for the calculation of the variation in body weight was:
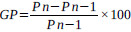
|
Where, GP = weight gain (%)
Pn = nth measure of body weight
Pn-1 = (n-1)th measure of body weight
2.7. Macroscopic Analyses of Organs and Biochemical Analysis of Blood
The animals were made to fast during the night preceding blood collection. They were then anesthetized using 0.5 mL of thiopental and blood samples were collected by cardiac puncture. The blood was collected in plain glass tubes for serum and centrifuged at 3000 rpm for 5 minutes to obtain serum. The liver, the heart, the kidney, the spleen and the lung were collected, rinsed in saline solution (0.9% NaCl), pressed on a filter paper and weighed using an electronic balance. The relative weight of each organ was evaluated using the following formula:
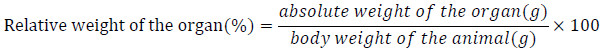
|
The serum obtained was used for biochemical measurements, carried out according to protocols provided in commercial kits (Biolab, Cat 150515 FRA). It includes Alanine Aminotransferase (ALT) and aspartate aminotransferase (AST). Assays were conducted for glucose, triglycerides, urea, creatinine (RECKON DIAGNOSTICS kit. Cat 100713 USA), total proteins and total cholesterol (BIOSYSTEMS kit). Hematological parameters were determined using an automated hematology analyzer (Hospitex Diagnostics Hema 18 Samsung 2000) and included blood count (red blood cells, white blood cells, hemoglobin, hematocrit, mean corpuscular hemoglobin, corpuscular hemoglobin concentration, and platelets).
2.8. Statistical Analysis
The data were processed by R software version 3.2.0 and Microsoft Excel version 2010. Statistical comparisons were made using one-way ANOVA and post ad hoc tests for specific comparisons. The significance of the differences between the treated and control groups was evaluated by Student's t-test, the differences were considered significant for p <0.05.
3. RESULTS
3.1. Main Chemical Groups Highlighted in Extracts by Characterization Reactions
3.1.1. Antiplasmodial Activity
Tables 2 and 3 show the effects of A. bipindensis and S. siamea extracts on the parasitemia of mice. Extracts of A. bipindensis at 180, 360 and 720 mg/kg and S. siamea at 100, 200 and 400 mg/kg showed antiplasmodial activity greater than 50% in all BALB / c mice infected with Plasmodium berghei with a significant difference (p <0.001).
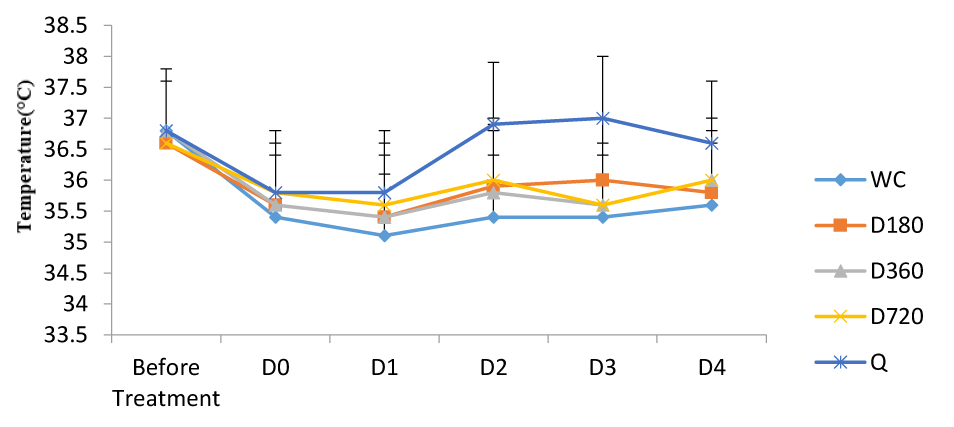
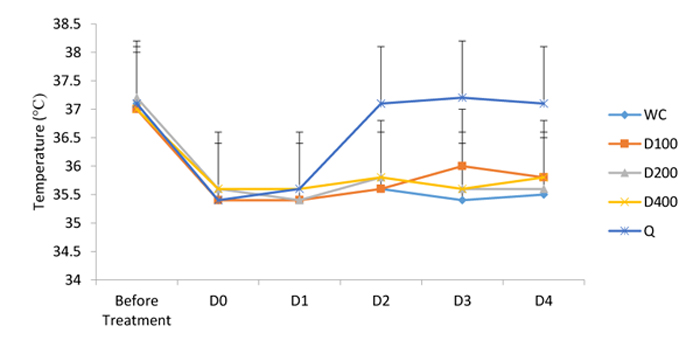
3.2. Effects of A. bipindensis and S. siamea Extracts on the Temperature of Parasitized Mice
Figures show the effect of A. bipindensis Fig. (2) and S. siamea Fig. (3) extract on the temperature of parasitized mice. the quinine-treated mice promptly increase in body temperature, whereas the other treatments, including the water negative control, stay low. However, the differences observed are not statistically significant.
| Extraction Solvents | Chemical Groups | Plant Drugs | |
| Leaves of Afzelia bipindensis | Leaves of Senna siamea | ||
| Ethanol-Water (70: 30%) | Phenolic compounds |
+++ | +++ |
| Tannins Catechin |
+ | - | |
| + | + | ||
| alkaloids | +/- | +/- | |
| Flavonoids |
+ | + | |
| Glucosides | - | + | |
| Steroids Triterpenes |
+ | - | |
| - | + | ||
| Saponosides | - | + | |
| Coumarins | - | +++ | |
| Treatment | Dose mg ̷Kg | Mean ±SD | Parasite Difference % |
| Extract | 180 | 25.96 ± 1.69 *** | 53.01±2.39 c |
| 360 | 24.97 ± 0.64 *** | 54.81±1.91 c | |
| 720 | 17.40 ± 0.62 *** | 68.50±1.49 b | |
| Quinine | 10 | 0.21 ± 0.14 *** | 99.61±0.17 a |
| Distilled water | 200µL | 55.24 ± 2.64 |
| Treatment | Dose mg ̷Kg | Mean ±SD | Parasite Difference % |
| Extract | 120 | 29.44 ± 2.23 *** | 46.70± 4.20 c |
| 240 | 20.21 ± 2.15 *** | 63.41± 2.97 b | |
| 480 | 18.54 ± 1.13 *** | 66.43± 2.26 b | |
| Quinine | 10 | 0.21 ± 0.14 *** | 99.61± 0.17 a |
| Distilled water | 200µL | 55.24 ± 2.64 |
3.3. Toxicity Observations and Effects of A. bipindensis and S. siamea Extracts on the Eeight
The observations made on animals in acute tests. There was no change in the physical appearance and somatomotricity of the animals during the observation period. No manifestations of tremor, seizures, salivation, diarrhea, coma, or abnormal behaviours such as self-mutilation or walking backward were observed for both extracts. Lethargy was observed in all animals in the first 30 minutes after force-feeding.
The effect of A. bipindensis and S. siamea on the weight of the mice was also evaluated. The difference in the body weight of the rats was statistically insignificant. However, the S. siamea treated rats showed less weight gain compared to the control rats.
3.4. Effects of A. bipindensis and S. siamea Extracts on Eaematological and Eiochemical Parameters
Tables ( 4-7) show the effect of extracts on hematological and biochemical parameters. For A. bipindensis we found a statistically significant difference in triglyceride levels between the water control group and the D360 and D720 groups. There is also a significant difference between ALT at D720 compared with other groups. Regarding Senna siamea, no statistically significant differences in the haematological and biochemical parameters between the different groups of animals were observed.
3.5. Effects of Afzelia bipindensis and Senna siamea Extracts on the Relative Weight of Organs
Tables 8 and 9 show the effect of extracts of Afzelia bipindensis and Senna siamea on the relative weight (%) of some organs. The values in the tables represent the averages of the relative weights of each organ as a function of dose; there is no significant difference between groups regardless of the extract.
Table 4.
| Hematological Parameter | Control | Dose 180 mg/kg | Dose 360 mg/kg | Dose 720 mg/kg |
| Leucocytes (103/mm 3) | 7.66 ± 4.66 | 5.30 ± 2.34 | 7.50 ± 2.96 | 6.62 ± 1.10 |
| Erythrocytes (106/mm 3) | 5.06 ± 3.04 | 3.80 ± 1.22 | 5.42 ± 0.93 | 6.32 ± 0.70 |
| Hemoglobin (g/dl) | 10.46 ± 5.11 | 8.99 ± 6.04 | 10.66 ± 2.23 | 10.30 ± 1.50 |
| Hematocrit (%) | 30.92 ± 15.05 | 35.77 ± 10.22 | 30.90 ± 6.33 | 35.15 ± 6.50 |
| Platelets (103/mm 3) | 229.40 ± 36.41 | 233 ± 42.46 | 252.70 ± 34.67 | 287.32 ± 56.44 |
| MCV (fl) | 87.00 ± 12.43 | 78.40 ± 15.40 | 90.70 ± 2.98 | 82.60 ± 5.96 |
| MCHT (pg) | 29.62 ± 1.17 | 35.74 ± 3.84 | 34.66 ± 1.44 | 34.68 ± 2.56 |
| MCHC (g/dl) | 33.98 ± 1.09 | 31.45 ± 0.39 | 34.06 ± 0.44 | 36.39 ± 0.88 |
| Hematological Parameter | Control | Dose 100 mg/kg | Dose 200 mg/kg | Dose 400 mg/kg |
| Leucocytes (103/mm 3) | 7.66 ± 4.66 | 4.38 ± 3.46 | 7.70 ± 3.93 | 5.82 ± 1.20 |
| Erythrocytes (106/mm 3) | 5.06 ± 3.04 | 4.83 ± 1.37 | 3.95 ± 0.85 | 4.44 ± 0.63 |
| Hemoglobin (g/dl) | 10.46 ± 5.11 | 9.72 ± 7.66 | 11.54 ± 2.79 | 11.40 ± 1.91 |
| Hematocrit (%) | 30.92 ± 15.05 | 31.76 ± 12.61 | 32.88 ± 8.14 | 34.10 ± 5.51 |
| Platelets (103/mm 3) | 229.40 ± 36.41 | 220.20 ± 32.22 | 242.40 ± 42.79 | 247.20 ± 46.44 |
| MCV (fl) | 87.00 ± 12.43 | 89.40 ± 9.88 | 82.80 ± 2.77 | 76.80 ± 7.36 |
| MCHT (pg) | 29.62 ± 1.17 | 24.94 ± 0.84 | 27.06 ± 0.90 | 25.68 ± 2.97 |
| MCHC (g/dl) | 33.98 ± 1.09 | 30.28 ± 3.24 | 35.06 ± 0.29 | 33.34 ± 0.73 |
| Biochemical Parameters | Control | Dose 180 mg/kg | Dose 360 mg/kg | Dose 720 mg/kg |
| AST | 64.00±5.24 | 65.22±17.34 | 64.32±18.30 | 60.26±10.44 |
| ALT | 140.85±17.36 | 160.93±12.12 | 171.38±16.25 | 213.58±20.50* |
| Glucose | 63.56 ±17.96 | 78.64 ± 22.40 | 64.24 ± 19.50 | 80.28 ± 20.98 |
| Blood urea nitrogen | 65.02 ± 11.61 | 64.44 ± 14.74 | 70.22 ± 16.11 | 74.90 ± 10.60 |
| Total Protein | 90.77 ± 22.70 | 87.07 ± 16.35 | 88.79 ± 23.82 | 85.76 ± 10.23 |
| Triglycerides | 229.42 ± 29.22 | 289.48 ± 29.89 | 123.89 ± 37.09* | 119.38 ±22.75* |
| Total Cholesterol | 65.67 ±10.55 | 60.31 ± 14.20 | 74.23 ± 17.80 | 71.89 ± 11.16 |
| Creatinine | 0.88 ± 0.54 | 2.03 ± 1.36 | 0.79 ± 0.39 | 0.84 ± 0.30 |
| Biochemical Parameters | Control | Dose 100 mg/kg | Dose 200 mg/kg | Dose 400 mg/kg |
| AST | 64.00±5.24 | 57.71±11.60 | 54.37±7.53 | 50.40±10.88 |
| ALT | 140.85±17.36 | 174.40±20.59 | 168.42±14.46 | 170.11±14.96 |
| Glucose | 63.56 ±17.96 | 76.93 ± 24.68 | 79.20 ± 18.54 | 72.27 ± 16.93 |
| Blood urea nitrogen | 65.02 ± 11.61 | 73.70 ± 17.34 | 77.31 ± 18.21 | 80.37 ± 13.68 |
| Total Protein | 90.77 ± 22.70 | 89.04 ± 12.58 | 78.59 ± 23.17 | 85.71 ± 27.89 |
| Triglycerides | 229.42 ± 29.22 | 248.48 ± 39.89 | 223.89 ± 24.56 | 220.21 ±18.70 |
| Total Cholesterol | 65.67 ±10.55 | 78.21 ± 13.24 | 74.01 ± 7.89 | 74.99 ± 11.13 |
| Creatinine | 0.88 ± 0.54 | 1.41 ± 1.88 | 0.80 ± 0.37 | 0.89 ± 0.32 |
| Organ | Control | Dose 180 mg/kg | Dose 360 mg/kg | Dose 720 mg/kg |
| Heart | 0.47±0.04 | 0.42±0.04 | 0.41±0.06 | 0.36±0.04 |
| Liver | 4.10±0.44 | 4.02±0.53 | 4.33±0.53 | 3.69±0.24 |
| Lungs | 0.80±0.10 | 0.80±0.14 | 0.89±0.25 | 0.96±0.18 |
| Kidney | 0.94±0.14 | 0.88±0.06 | 0.90±0.04 | 0.97±0.14 |
| Spleen | 0.61±0.10 | 0.62±0.11 | 0.52±0.09 | 0.60±0.24 |
| Organ | Control | Dose 100 mg/kg | Dose 200 mg/kg | Dose 400 mg/kg |
| Heart | 0.47±0.04 | 0.44±0.07 | 0.43±0.04 | 0.38±0.06 |
| Liver | 4.10±0.44 | 3.77±0.40 | 3.51±0.56 | 3.74±0.33 |
| Lungs | 0.80±0.10 | 1.14±0.51 | 1.06±0.23 | 1.07±0.36 |
| Kidney | 0.94±0.14 | 0.97±0.14 | 0.93±0.04 | 0.86±0.11 |
| Spleen | 0.61±0.10 | 0.57±0.08 | 0.53±0.12 | 0.56±0.10 |
4. DISCUSSION
Malaria is a serious disease and its prevalence is increasing across tropical countries. For many decades, traditional medicine has been used in the treatment of many diseases including malaria. This study evaluated the antimalarial effect of Afzelia bipindensis and Senna siamea extracts. Afzelia bipindensis and Senna siamea extracts were found reducing malaria in white BALB/c mice. This study helped us show that hydroethanolic extracts of Afzelia bipindensis and Senna siamea reduce to more than half the parasitemia in murine models. The two plants showed good efficiency (Tables 1 and 2) against the Plasmodium berghei.
In fact, the antimalarial activity observed could be attributed to the secondary metabolites present in Afzelia bipindensis and Senna siamea, moreover these plants are a rich source of alkaloid, phenolic compound, and flavonoids which contribute in their antiplasmodial activity [16-20].
Phytochemical screening of Senna siamea essential oil equally reveals the presence of pure components such as limonenes [21] with an in vitro antiplasmodial activity against FcB1 Plasmodium falciparum with an IC50 varying between 0.5 and 9.8 µg/ml [22].
According to the review covered and to the limits of our knowledge, few studies have been published on the in vivo antiplasmodial activity of Afzelia bipindensis and Senna siamea.
In this study, all treatments induce a reduction in temperature, the quinine-treated mice promptly increase in body temperature, whereas the other treatments, including the water negative control, stay low. One of the major characteristics of malaria is the presence of a swaying fever, and quinine can thwart the polymerisation of heme from haemoglobin and hinder from here the reproduction of the Plasmodium parasite, thus limiting the liberation of pyrogenic substances.
The acute toxicity evaluation of hydroethanolic extracts of Afzelia bipindensis and Senna siamea in mice can be helpful for their use in traditional medicine to treat malaria in human. The only modifications observed due to the treatment occurred at the first hours of administration of the extract (a little more than four hours for animals treated with Senna siamea and two hours for those treated with Afzelia bipindensis). The animals were dizzy, but these symptoms were fleeting. No other occurrence and weight loss linked to the treatment were observed. These results suggest that these extracts present fair risks for health in unique doses.
According to the review covered, and to the limits of our knowledge, a few studies were carried out on the sub-acute toxicity of Afzelia bipindensis and Senna siamea. To identify the risks against human health due to repeated intake of extracts, a sub-acute toxicity test was carried out during the repeated oral administration of extracts at 180, 360 and 720 mg/kg doses for Afzelia bipindensis and at 100, 200 and 400 mg/kg doses for Senna siamea in a period of 28 days. No treatment-linked modification was found in animals during the whole period of the treatment. No significant weight loss was recorded in the animals for the different extracts compared to the controls.
Senna siamea extracts brought no significant modification of biochemical parameters evaluated (glucose, total protein, Blood urea nitrogen, total cholesterol and creatinine) and haematological (Numerations and blood count), same observation for Afzelia bipindensis extract at all doses; meanwhile we noted an increase in AST level for the 720 mg/kg dose compared to the control group; this level remained normal for the other doses. We also have an increase in the level of serum triglycerides for doses 360 and 720 mg/kg compared to the control group; this level remained normal for other doses. An increase in serum AST can signify a cardiac dysfunction, hepatopathies, muscular dystrophia and damage to internal organs.
An autopsy of the animals revealed a general darkening of the liver with the presence of inflammatory nodules in animals treated with Afzelia bipindensis at 720 mg/kg dose and two animals treated with Senna siamea at 200 mg/kg and 400 mg/kg doses respectively compared to the controls. Control of the thoracic cavity revealed the presence of inflammatory nodules of the lung of an animal treated with Afzelia bipindensis at 360 mg/kg dose and an animal treated with Senna siamea at 200 mg/kg dose compared to the controls. The inflammatory nodules of the liver and lung observed were similar. If the treatment continues for over a long duration, the nodules will be detected in more animals and will subsequently lead to organs deficit such as necrosis, sclerosis cirrhosis and cancer.
The kidney of an animal treated with Senna siamea at 400 mg/kg dose was atrophied. The other organs of the animals presented no distinctive signs compared to the control animals. Nevertheless, we noted no statistically significant difference between the relative weights of all the animals treated compared to the control.
CONCLUSION
The study showed that extracts of Afzelia bipindensis and Senna siamea significantly reduce plasmodium activity in mice. In addition, extracts of these plants at tested doses showed no health risk, thus supporting the use of these compound in the treatment of malaria. However, studies with other Plasmodium strains (Plasmodium falciparum) are suggested.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Animal Care and Use Committee of the Institute of Medical Research and Medical Plants Studies in Cameroon.
HUMAN AND ANIMAL RIGHTS
Humans did not participate in this research. All animal research procedures followed were in accordance with the US National Research Council's “Guide for the Care and Use of Laboratory Animals”.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared None.


