All published articles of this journal are available on ScienceDirect.
Synthesis of Some 4,5-Dihydrothieno[3,2-e][1,2,4]Triazolo[4,3-a] Pyrimi-dine-2-Carboxamides as Anti-Inflammatory and Analgesic Agents
Abstract
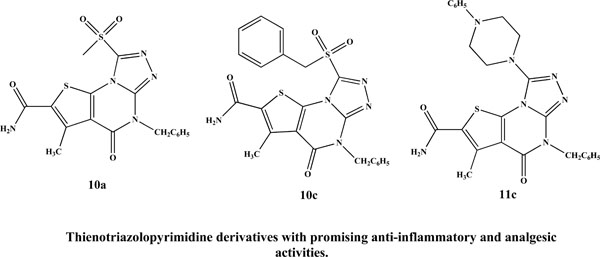
A new series 4,5-dihydrothieno[3,2-e][1,2,4]triazolo[4,3-a]pyrimidine-2-carboxamide was synthesized. Twenty one newly synthesized compounds were investigated for their anti-inflammatory and analgesic activity using acute and subacute formalin-induced paw edema models and diclofenac Na as a reference. The acute toxicity (ALD50) and ulcerogenic effects of the active compounds were also determined. The thienotriazolopyrimidines 10a, 10c and 11c were found to exhibit remarkable anti-inflammatory activity at both models in addition to good analgesic activity with a delayed onset of action. Moreover, the active compounds showed high GI safety level and are well tolerated by experimental animals with high safety margin (ALD50 > 0.4 g/kg). Docking study using Molecular Operating Environment (MOE) version 2008.10 into COX-2 has been made for derivatives of highest anti-inflammatory activity.